Understand the calculation of anodes for ships: How to protect your ship from corrosion
Learn the importance of sacrificials for vessels and how our services at Anodefactory can help you calculate your specific need.
In order to keep your ship safe from corrosion and extend its life, it is important to use sacrificials. Operaranodes are made of a metal that is more reactive than the metals in your ship's hull and drive system, and they work by corroding before the rest of the ship's metal components. But how do you know what size and quantity of anodes your ship needs? It is about making a correct calculation & design of the system design. In this blog post, we will explore the importance of sacrificials for ship maintenance and how to correctly calculate the number and size of zinc anodes needed for your ship.
Why is for example. Zinc anodes important for vessels? Even in salt water environments, where corrosion is more likely to occur, the use of zinc anodes can protect your vessel from corrosion. Zinc anodes act as a victim metal that corrodes instead of your ship's metal components. Without zinc anodes, your ship's hull and drive system would be more susceptible to corrosion and damage, which could ultimately jeopardize the safety and life of your ship. Calculation of zinc anodes for vessels to protect your vessel with zinc anodes, you need to correctly calculate the correct size and number of anodes needed for your ship. How to use:
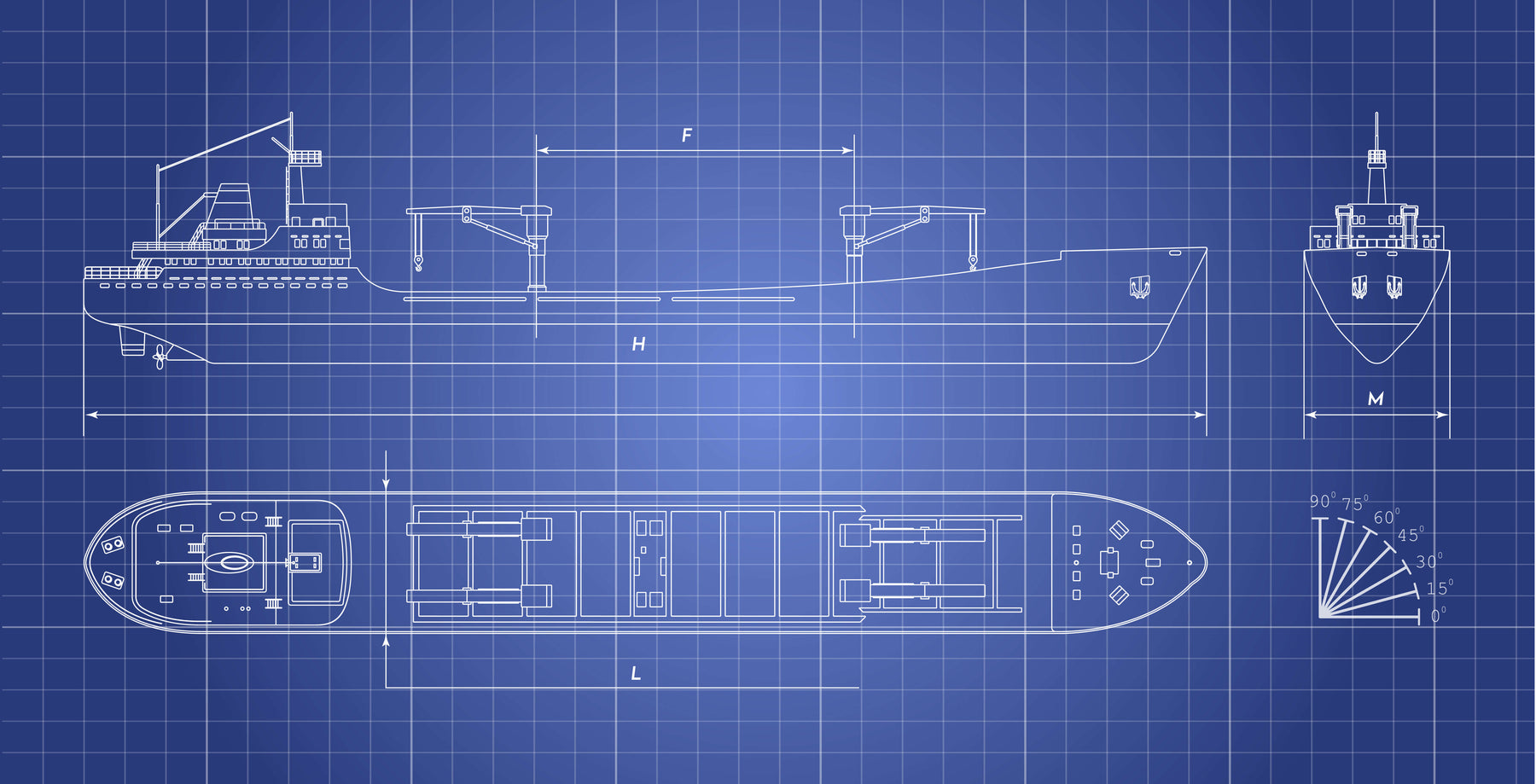
- Determine the surface of your ship's hull and drive system in M2.
- Choose the size of your ship and order a design of us
- Get a complete Anodplan showing how much and where to place your sacrificials.






























































Leave a comment